Detect Phase Separation before it impacts your business
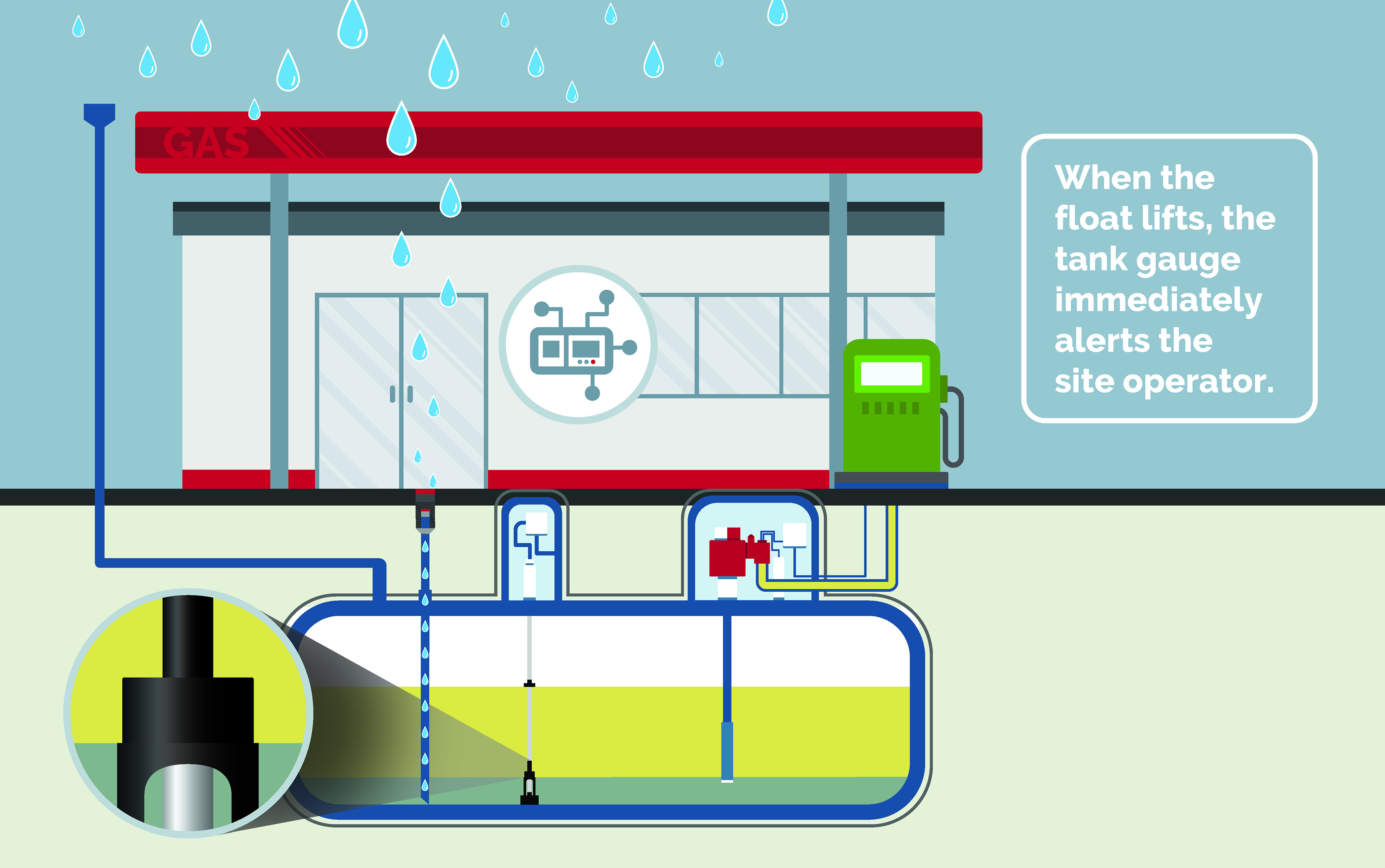
Early Detection
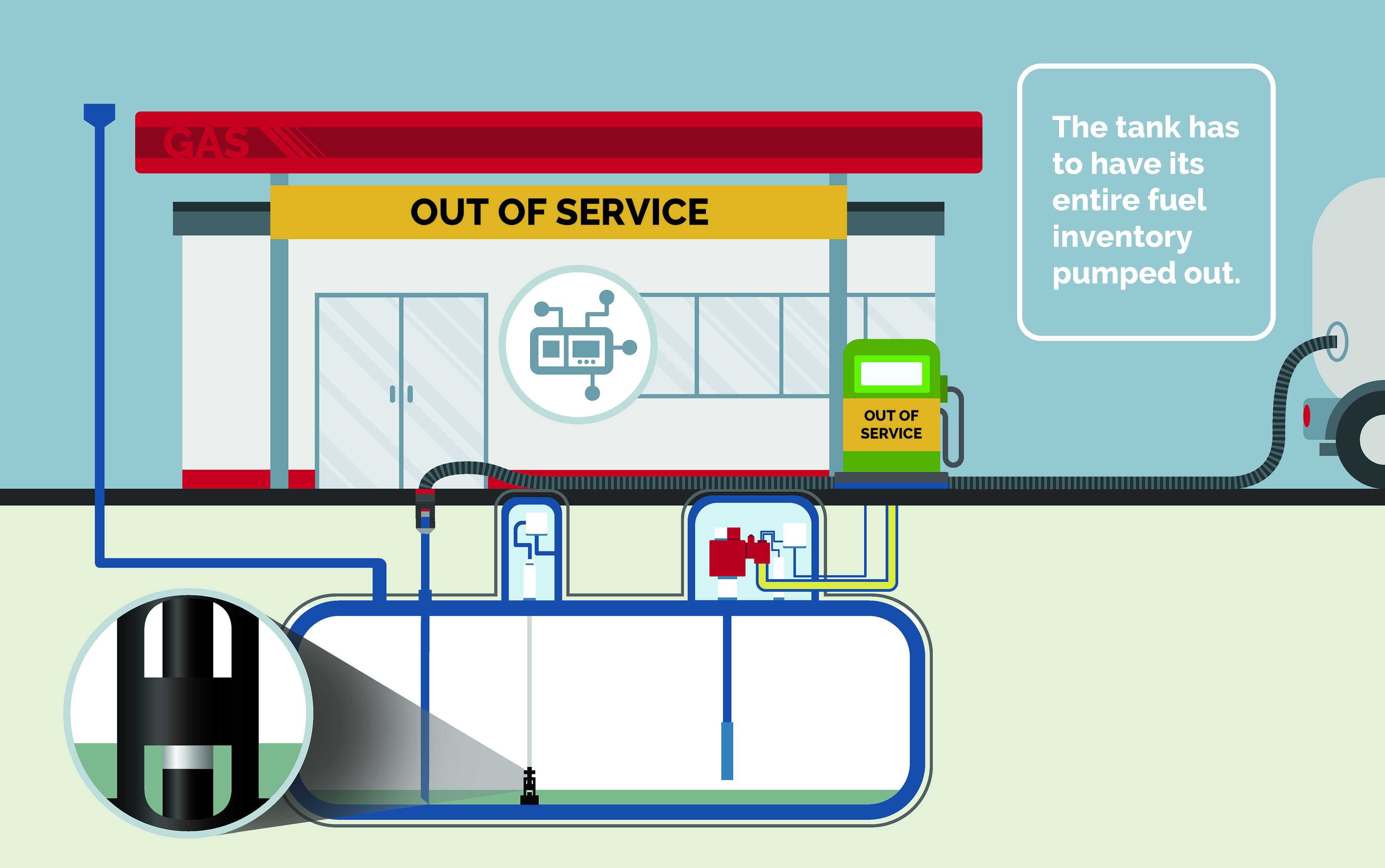
No customers or fuel lines have been affected. The phase separation may be resolved with a normal delivery of fuel. The site operator will need to pump out phase separation from the bottom of the tank and check octane levels of the remaining fuel.
Cost: Lost gallons of fuel, service, and lost business
Clogged Filters
If phase separation has clogged the dispenser fuel filters, the remaining fuel is ethanol depleted and may be out of specification. The site operator may need to purge lines and change filters or possibly pump out the entire tank of fuel. The tank will be down for a few hours.
Cost: Lost tank of fuel, service & repairs, and lost business.
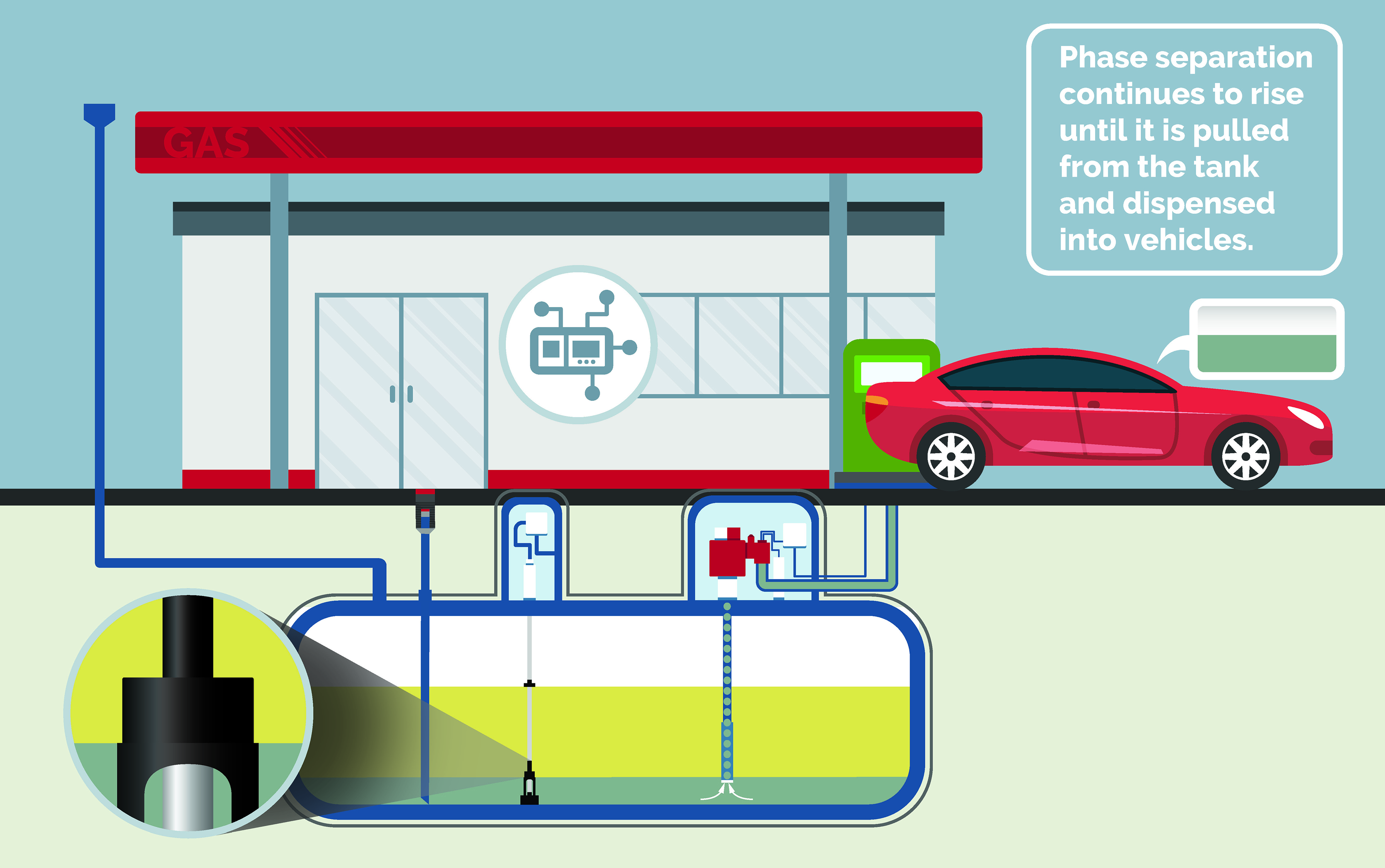
Stalled Vehicle
Vehicles stalled out at the station, resulting in legal liabilities and a damaged reputation. The site operator will need to purge lines, change filters, and pump out the entire tank of fuel. They will also need to repair damaged vehicles and begin damage control for their brand.
Cost: Lost tank of fuel, service & repairs, lost business, and damage to your reputation.
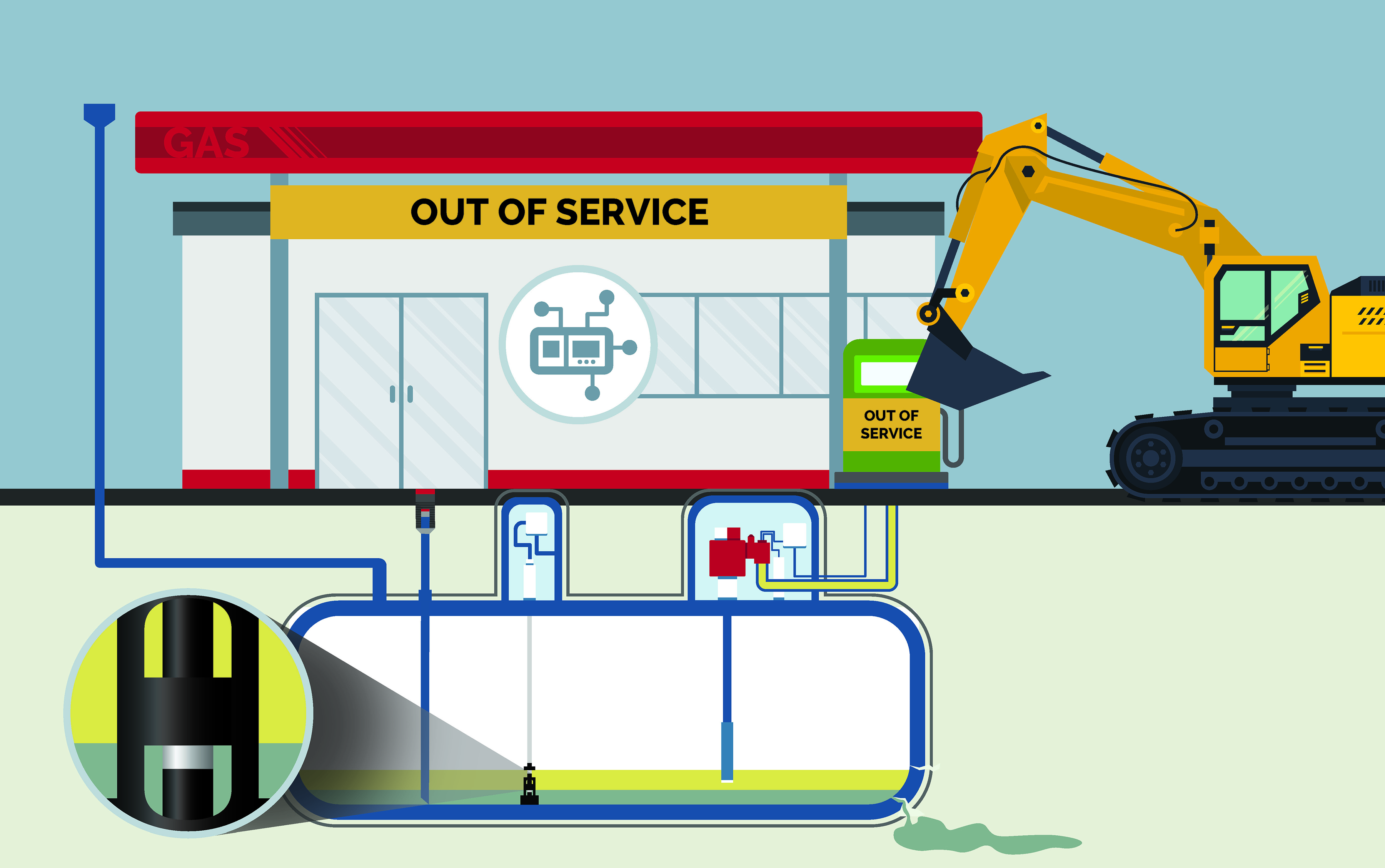
Corroded Tanks
Tanks and piping can start leaking with prolonged phase separation exposure. Now the site operator will need to extract underground tanks and piping, as well as clean up leaking gas.
Cost: Potential shutdown, fines and Notice of Violations, lost tank of fuel, service & repairs, lost business, and damage to your reputation.
What can you do about it?
The only way to combat phase separation is with continuous monitoring and early detection. The Veeder-Root Phase Separation Float Kits replace the traditional gasoline float kit, offering both a water float and a phase separation float that send alarms straight to your automatic tank gauge when tank contamination is suspected.

 Ethanol usage is on the rise, and subsequently so is Phase Separation
Ethanol usage is on the rise, and subsequently so is Phase Separation